
When one mole each of gaseous Na + and Cl – ions form solid NaCl, 769 kJ of heat is released. Thus, it requires 769 kJ to separate one mole of solid NaCl into gaseous Na + and Cl – ions.
For sodium chloride, Δ H lattice = 769 kJ. In both cases, a larger magnitude for lattice energy indicates a more stable ionic compound. Thus, make sure to confirm which definition is used when looking up lattice energies in another reference. Another way is to use an equivalent, but opposite convention, wherein the lattice energy is exothermic (negative values) and described as the energy released when ions combine to form a lattice. Here, the convention is used where the ionic solid is separated into ions, meaning the lattice energies will be endothermic (positive values). The lattice energy (Δ H lattice) of an ionic compound is defined as the energy required to separate one mole of the solid into its component gaseous ions.

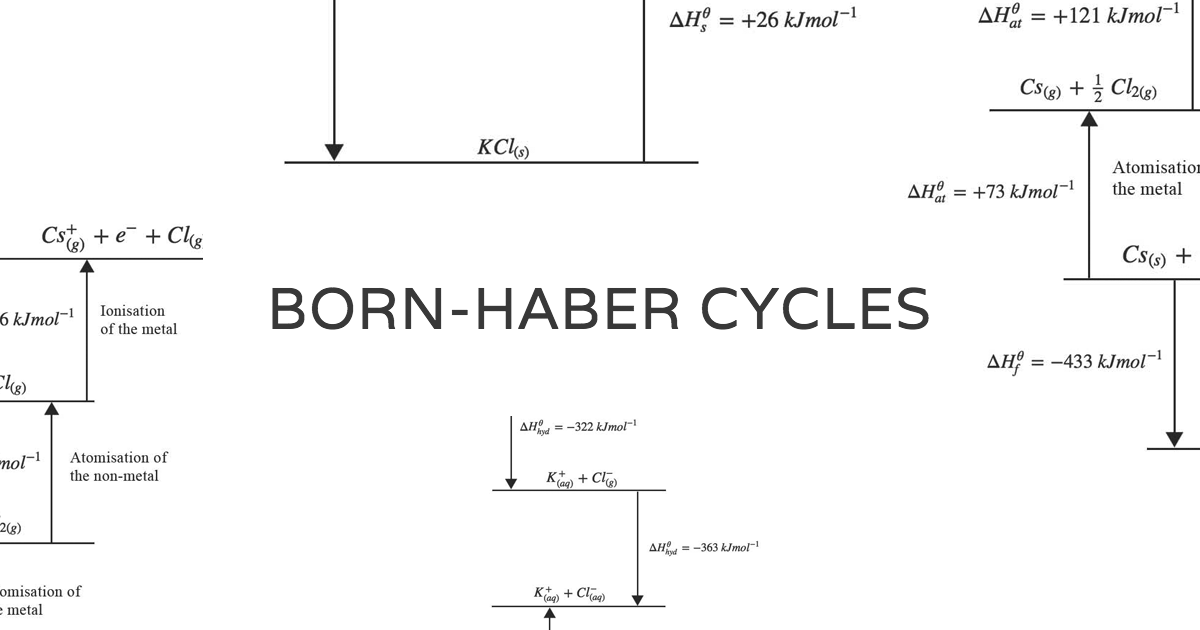
The lattice energy of a compound is a measure of the strength of this attraction. Meaning the enthalpy value of the direct route equals the sum of enthalpies of the five steps.īy solving the equation for lattice energy, a large negative value is determined, which signifies an exothermic reaction.Īn ionic compound is stable because of the electrostatic attraction between its positive and negative ions. Hess’s law states that the change in the overall enthalpy of a stepwise process is the sum of the enthalpy changes of each step. In the final step, the electrostatic attraction between the gaseous ions leads to the formation of the lattice structure. The electron is then taken up by the gaseous chlorine to form a chloride anion. An electron is removed from the gaseous sodium to form a sodium cation. The third and fourth steps account for the electron transfer to form ions. Next, diatomic chlorine molecules dissociate into gaseous chlorine atoms. In the first step, solid-state sodium is converted into its gaseous form. The direct route represents the standard enthalpy of formation of NaCl from elemental sodium and chlorine.
Born haber cycle problems with answers series#
However, it can be calculated using Hess’s law in a hypothetical series of steps called the Born–Haber cycle, which represents the formation of an ionic compound from its constituent elements.įor example, the Born-Haber cycle for sodium chloride formation considers two alternate routes, one direct and another indirect. In a solid ionic compound, a large number of charged particles interact with each other making it difficult to determine the exact value of the lattice energy experimentally. The total energy associated with the formation or breakdown of a crystalline lattice into its gaseous constituents is called lattice energy. The resulting lattice structure is stabilized by decreasing the potential energy, which is released as heat, an exothermic reaction. But where is the energy coming from?Īccording to Coulomb’s law, cations and anions are attracted to each other by strong electrostatic forces into a solid array or lattice. However, when elemental sodium and chlorine react to form a solid sodium chloride crystal, it's a highly exothermic process. Pressing the "Show Answer" button will cause the correct answer to display and you will no longert be able to submit an anser for that question.Formation of ionic bonds requires an electron transfer from a metal to a nonmetal atom - a process that is often endothermic.Nonetheless, the only way to get the "correct answer" is to do a proper Born-Haber analysis of the data provided. In most cases, the change is less than 1%. To remove the possibility of your "answering" the questions by merely looking in a reference book, the thermodynamic values the page presents have been very slightly altered from the literature values.The page creates and calculates the lattice energy fresh each time.The data is presented in such a fashion that all answers calculated should be integers and should be negative. Be very careful with your calculations.There are a number of very important rules: Regardless, determine the value of the missing heat, enter it and press "Check Answer". Most of the time, the missing heat will be the lattice energy, but, occasionally, it will be another term. When you press "New Problem", a window will open with a set of thermochemical data related to an ionic compound. This page provides exercises in the use of the Born-Haber cycle to determine lattice energies of ionic compounds.
